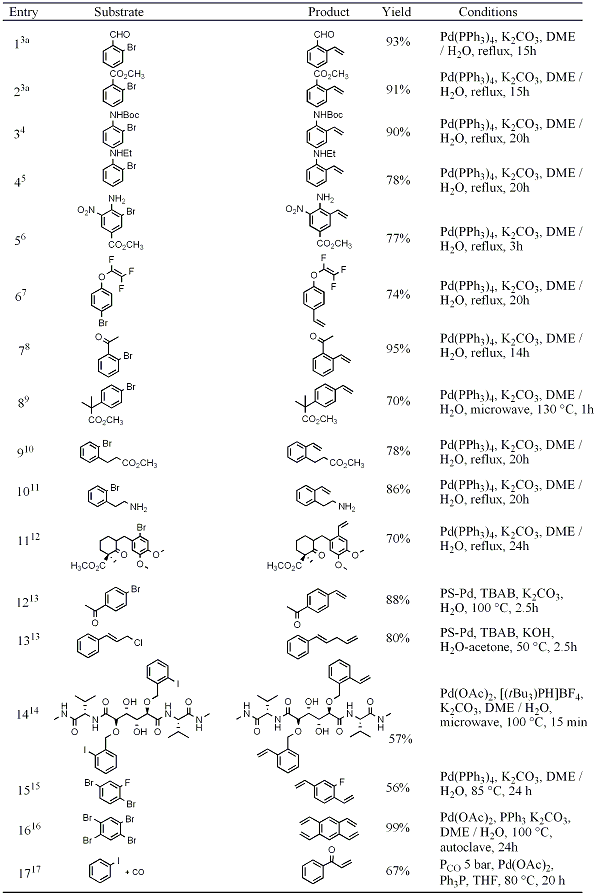
Enantioselective α-Vinylation of Aldehydes via the Synergistic Combination of Copper and Amine Catalysis | Journal of the American Chemical Society
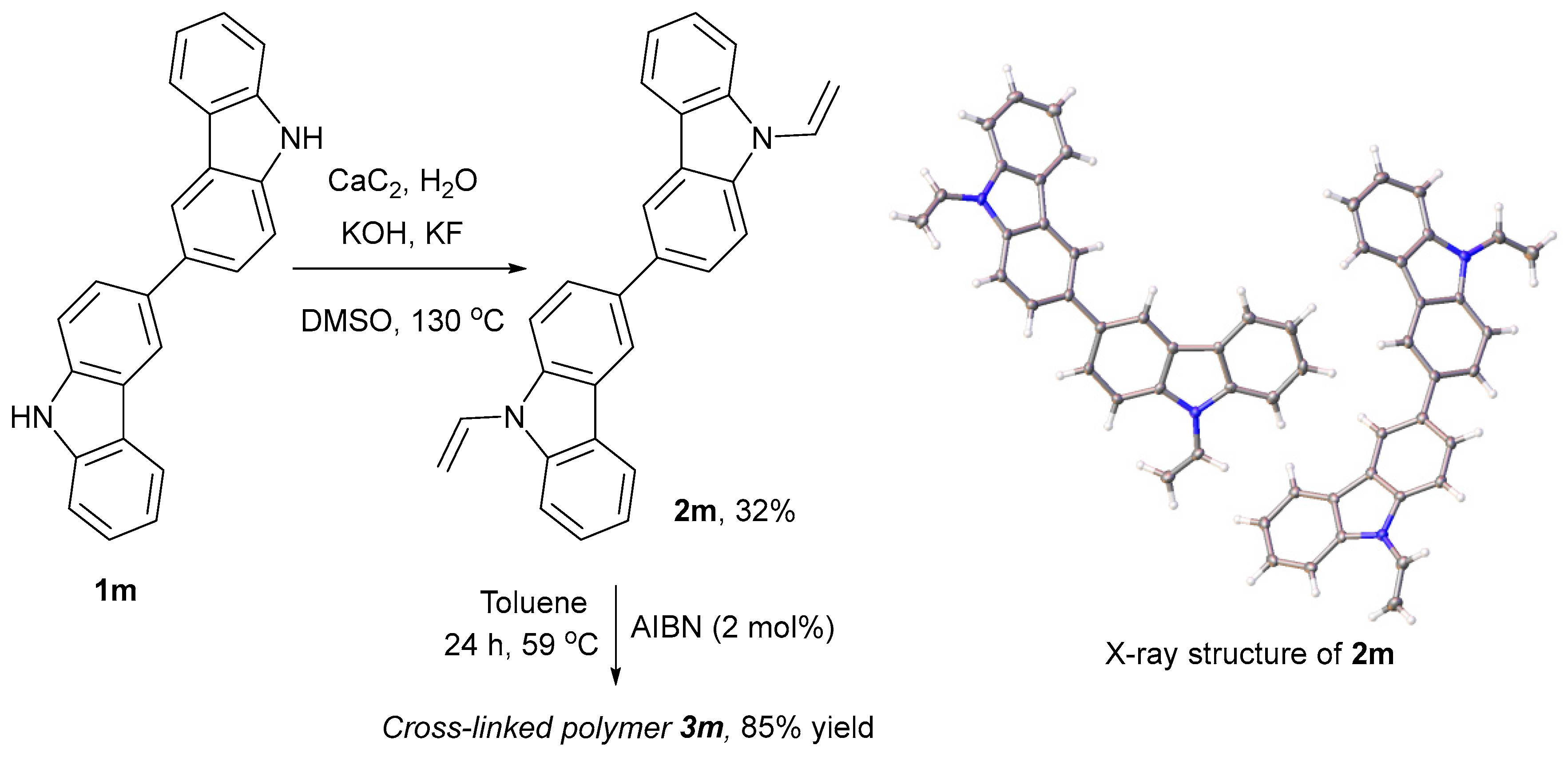
Molecules | Free Full-Text | Vinylation of a Secondary Amine Core with Calcium Carbide for Efficient Post-Modification and Access to Polymeric Materials
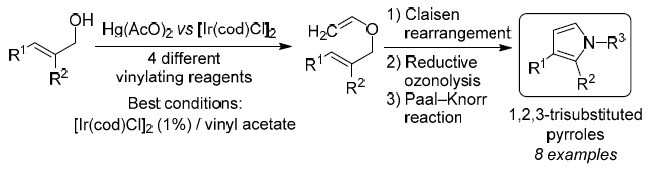
Synthesis of 1,3- and 1,2,3-functionalized pyrroles via Ir(I)-catalyzed vinylation of allyl alcohols | SpringerLink

Low pressure vinylation of aryl and vinyl halides via Heck–Mizoroki reactions using ethylene - ScienceDirect

Direct Vinylation of Alcohols or Aldehydes Employing Alkynes as Vinyl Donors: A Ruthenium Catalyzed C−C Bond-Forming Transfer Hydrogenation | Journal of the American Chemical Society

J. Am. Chem. Soc. on Twitter: "One of our most read articles in the last 30 days: Vinyl Thianthrenium Tetrafluoroborate: A Practical and Versatile Vinylating Reagent Made from Ethylene by @FabioJulia7, Jiyao

Pd‐Catalyzed Vinylation of Aryl Halides with Inexpensive Organosilicon Reagents Under Mild Conditions - Yang - 2018 - Chemistry – A European Journal - Wiley Online Library

Use of Iridium‐Catalyzed Transfer Vinylation for the Synthesis of Bio‐Based (bis)‐Vinyl Ethers - Spiegelberg - 2022 - Advanced Synthesis & Catalysis - Wiley Online Library
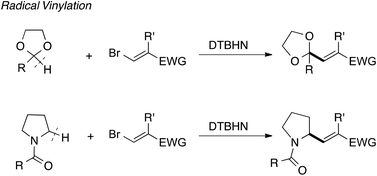
Radical vinylation of dioxolanes and N-acylpyrrolidines using vinyl bromides - Organic Chemistry Frontiers (RSC Publishing)

General Reaction Conditions for the Palladium-Catalyzed Vinylation of Aryl Chlorides with Potassium Alkenyltrifluoroborates
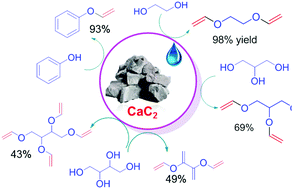
Direct vinylation of natural alcohols and derivatives with calcium carbide - Green Chemistry (RSC Publishing)
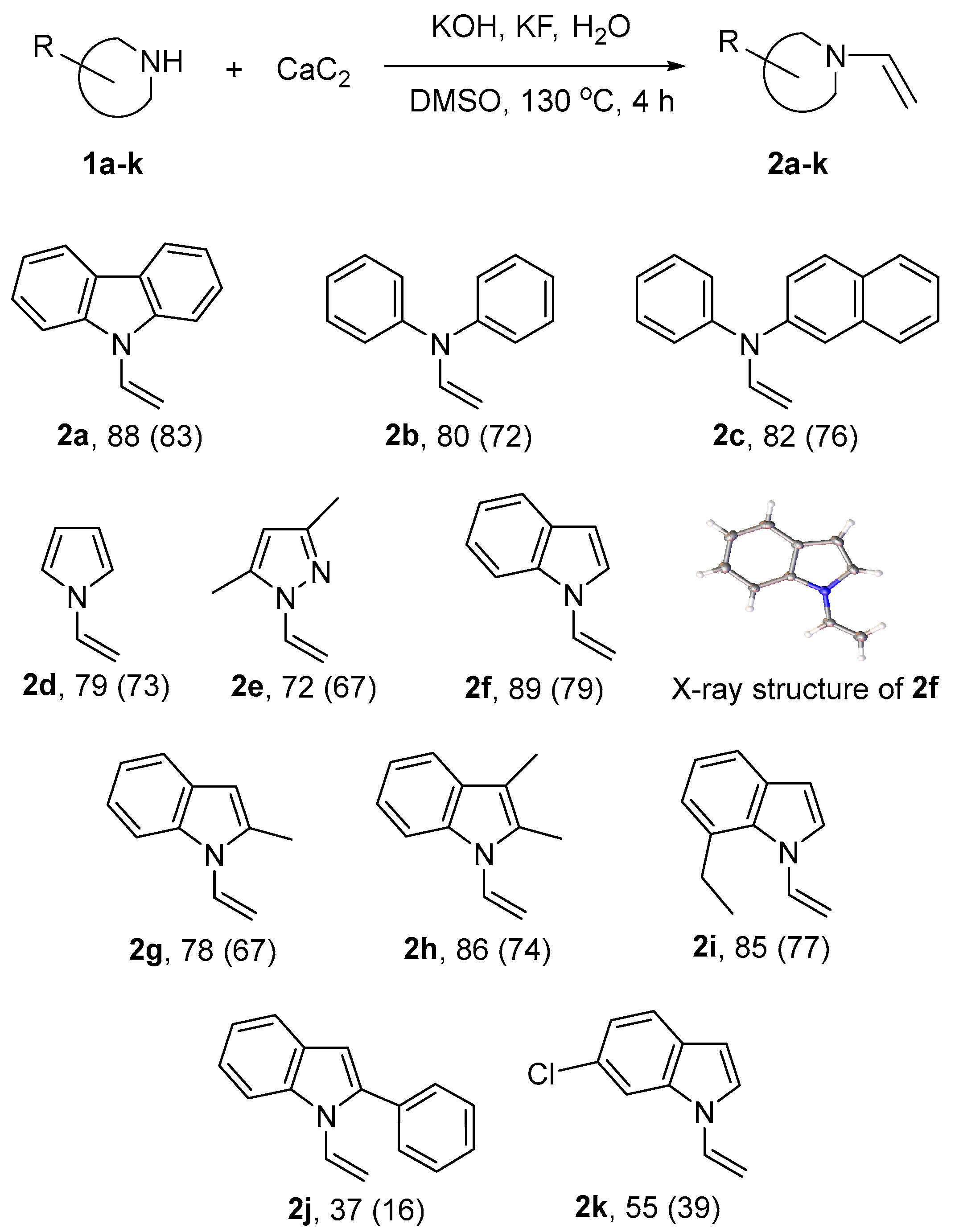
Molecules | Free Full-Text | Vinylation of a Secondary Amine Core with Calcium Carbide for Efficient Post-Modification and Access to Polymeric Materials

PDF) Studies of microwave-enhanced Suzuki–Miyaura vinylation of electron-rich sterically hindered substrates utilizing potassium vinyltrifluoroborate | Stefan M Cooper, Jr. - Academia.edu